hearing test
eAUDIOUSB
A new dimension in hearing measurement using pure tones and computer speech
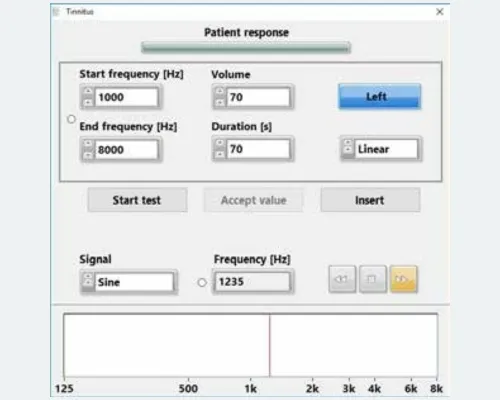
The eAUDIOUSB system: a new dimension in computer-assisted audiometry, encompassing both pure tones and speech. eAUDIOUSB is a state-of-the-art, computer-based audiometry system designed for everyday use. Utilizing the latest electronic technologies, eAUDIOUSB sets new standards in two-channel audiometry. Standardized charts are integrated into a clearly structured interface to ensure optimal system usability.
- 3 Active Free-Field Channels
- Patient Response
- USB 2.0 Port
- Elegant design suitable for wall or desk mounting
program eAUDIOUSB
The heart of the device beats within the computer. As part of the eAUDIO software, all functions are easily accessible. The device can be controlled manually or in a time-saving automatic mode. Using eAUDIOUSB, eABRUSB, and eOAE, a modern diagnostic center can be created, displaying all relevant data at a glance. The eAUDIO software is integrated with eDM – the Diagnostic Manager. Free online software updates are available.
| Probe Tone Frequencies | 226 Hz, 678 Hz, 1000 Hz ±1% (800 Hz @ 75 dB on request) |
| Test Tone Intensity | 85, 80, 75 dB SPL ±3 dB |
| Total Harmonic Distortion + Noise | Less than 4% (acoustic measurement) |
| Pressure Range | +400 ... -600 daPa |
| Pressure Accuracy | ±5% or 10 daPa |
| Compliance Range | 0.1 ... 6.0 ml (8.0 ml on request) |
| Compliance Accuracy | ±5% or 0.1 ml |
| Pump Speed | 100–400 daPa/sec |
| Pump Control | Automatic / Manual |
| Compliance Unit | Equivalent air volume [ml] or mmho |
| Number of Curves | One item per page with overwrite function |
| LED Function | Six colors and a flashing LED indicator to display the current status of the device and probe |
| IPSI Pure Tone | 500, 1000, 2000 and 4000 Hz ±3% |
| IPSI Indicator Intensity | Max. 110 dB HL ±3 dB |
| Pure Tone | 500, 1000, 2000 and 4000 Hz ±3% + user-defined stimulus |
| Contralateral Intensity | Max. 120 dB HL ±3 dB |
| Contra Headphone | DD45 Contra |
| THD+N | Less than 5% (acoustic measurement) |
| Minimum Intensity | 40 dB HL |
| Measurement | Automatic or Manual |
| Automatic Test | 5 dB / 10 dB steps per frequency |
| Manual Test | Unlimited number of curves per frequency and per ear |
| Stimulus Duration | 0.4 – 1.5 seconds (reflex decay 60 seconds) |
| Perforated Eardrum | Active and passive tube opening (Valsalva maneuver) |
| Intact Eardrum | Williams Test (3 curves per ear) |
| Dimensions (L × H × W) | 310 mm × 105 mm × 250 mm |
| Weight | Approx. 1500 g |
| Probe Weight | 4 g |
| Power Consumption | Max. 20 W |
| User Interface | Isolated USB 2.0 port |
| Test Types | Tympanometry, Acoustic Reflex Threshold, Reflex Decay, Eustachian Tube Function (intact and perforated) |
| Contra Output | 6.35 mm |
| Trigger Input | 3.5 mm, opto-isolated 5 kV, internally limited current from 5 to 20 mA |
| Play Output | 3.5 mm, opto-isolated 5 kV, open collector |
| Environment | 10°C to 40°C, max. humidity 90% |
| Standards | DIN EN 60645-5 - DIN EN 60601-1 - EWG 93/42 EEC |
Middle Ear Analysis
eTYMPUSB
Middle Ear Analyzer
The eTYMPUSB middle ear analyzer is a computerized middle ear analyzer designed for both routine and practical clinical use. The eTYMPUSB allows for the creation of customized test protocols by combining different tests from a suite into a single test path. For example, you can create a simple screening procedure and a more in-depth evaluation. The device features a compact, practical, and attractive design. Key features include: 226 Hz tympanic membrane measurement (standard), a small, lightweight, and easy-to-clean probe, various operating functions via signal separation, self- and contralateral reflex decay testing, Eustachian tube function (ETF) testing with intact and perforated tympanic membranes, measurement of the acoustic response to direct stimulation of a cochlear implant(CI), a scientific testing mode, automatic height correction for accurate permittivity values, storage of all relevant calibration values in the probe, and robust acoustic reflex detection.
Evoked Potentials
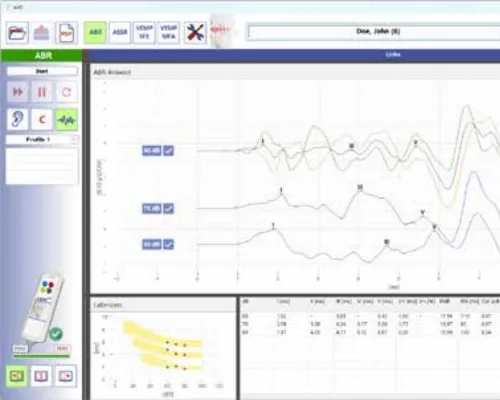
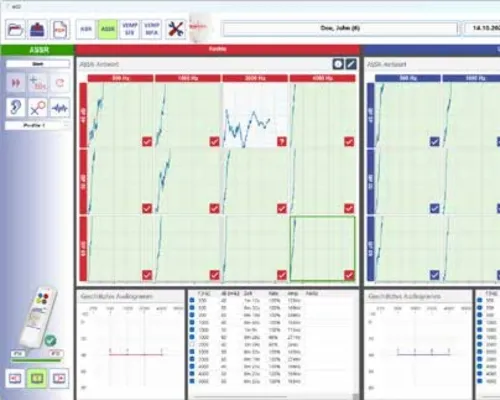
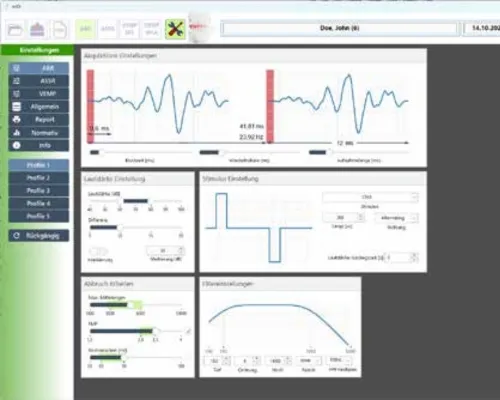
eABRUSB
The eABRUSB is a two-channel unit for measuring not only auditory evoked potentials (AEPs) but also vestibular evoked potentials (c- و oVEMP). Compact as a remote control, the eABRUSB is a fully integrated, portable device for measuring AEPs. Combining state-of-the-art electronics with a user-friendly interface, the eABRUSB ensures optimal results in both routine and clinical applications. It measures AEPs and vestibular evoked potentials (c- و oVEMP). The eABRUSB features integrated earphones and earpieces. The device automatically selects appropriate calibration values. The ability to create custom test protocols simplifies routine procedures. • Automated Assistance Functions: – Automatic electrode impedance control – Automatic gain control (AGC) – Tap, pulse, quick tap – Automatic noise rejection – Automatic shut-off function – Noise masking – Free selectable protocols – Easy-to-use curve manipulation – Automatic coupler detection – Free lifetime online subscription
Otoacoustic Emissions (OAE)
eOAE
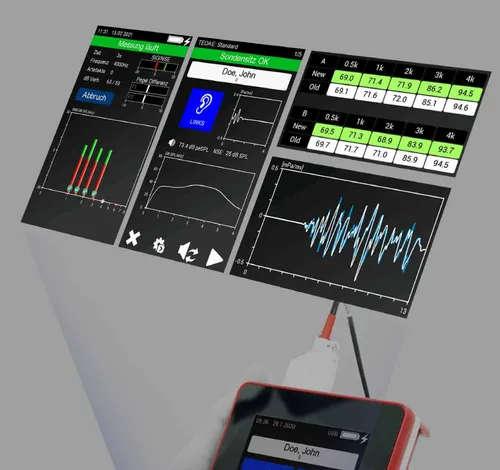
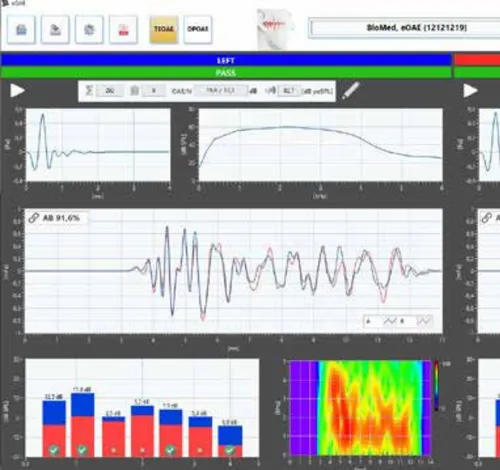
The eOAE portable device measures transient acoustic emissions (TEOAE) and distorted acoustic emissions (DPOAE): When an auditory signal reaches the auditory system, the inner ear emits a very faint sound known as otoacoustic emissions. Transient otoacoustic emissions (TEOAE) and distorted otoacoustic emissions (DPOAE) are distinguished. Both TEOAE and DPOAE can be measured using the eOAE device. A special screening mode is also available for newborn screening.
The eOAE device also measures transient otoacoustic emissions. It features: • Easy cleaning of probe parts • Good sterilizability thanks to user-defined stop parameters • 4 adjustable profiles • Touchscreen • Time graph or frequency chart display • Optional printer available • Clear management of measurement data on the device as well • QWERTZ keyboard for easy data entry on the device • Graph and tabular display • Full integration with eDM Diagnostic Manager • Easy charging via docking station • TEOAE method • PC software for remote operation • Clear display of results.
- Sampling rate: 48 kHz
- Analog-to-digital signal converter resolution: 24 bits
- Screen: 5-inch touchscreen
- Weight: 320 grams
- Battery: 3880 mAh, 400 tests minimum.
- Headphone output for coverage
- Dimensions: 141 x 97 x 27 mm
- Type: Thermal printer
- Paper width: 57.5 mm
- Resolution: 8 pixels/mm, 384 pixels/line
- TypeTEOAE وDPOAE
- The catalystTEOAE: Non-linear clicks
DPOAE: Pure tone f1:f2 = 1:1.2 - Probe cable length1.5 meters
- Frequency rangeTEOAE: 1-4 kHz
DPOAE: 0.5-12 kHz - LevelTEOAE: 40-90 dB peSPL
DPOAE: 40-70 dB SPL
AudioBox
The AudioBox is a 4- to 6-channel free-field amplifier. This modern, computer-controlled free-field amplifier is designed to connect up to six passive speakers. The six output channels can be allocated to four input channels. Each input channel can be individually calibrated to up to 50 positions (125 Hz, 250 Hz, etc.). Additionally, upon customer request, the device includes six signal channels for event display. The AudioBox can be used to extend the capabilities of a conventional audiometer for auditory aptitude analysis or pediatric audiometry. Technical specifications: • Windows software • Medical power supply • 4 inputs (maximum 5V) • 6 outputs (100W each) • Isolated USB 2.0 port • API for C/C++, C#, Delphi, and LabVIEW languages

AudioControl
Audiometry Keyboard
This keyboard is specifically designed to provide a comfortable experience during audiometry testing. Two sliders allow you to control tone volume and sound coverage. Sixteen touch-sensitive buttons ensure completely silent operation.
This keyboard works with eAUDIOUSB software and can be easily integrated into OEM applications via the HID Joystick interface.

eDM
Electronic Diagnostic Manager
The Electronic Data Management System (eDM) simplifies and streamlines daily operations.
The (eDM) is the ideal solution for measuring, displaying, managing, and storing ENT data obtained by BioMed Jena devices.
All different tests are easily accessible. To optimize workflow, a measurement schedule can be created for each patient. The (eDM) can also import PDF files from other sources.
Provide remote support with ease—launch the Anydesk remote control software directly from the (eDM).

Key Features of the Electronic Diagnostic Manager (eDM) Software
One Software for All BioMed Jena Devices
- A single patient database
- Easy and quick data preview
- Customizable display for all measurements
- Easy data management (one data for all)
- Import external PDF files / Automatically export PDF files
- User profiles with independent settings for each user.
Network support
- Create a measurement chart for each patient
- Firebird database with no storage restrictions
- No workplace license required
- GDT interface included, HL7 available upon request

For more information
Feel free to contact us anytime. Our team is always ready to help you.